New study confirms that purity and type of coating biomaterials used in 2D cell culturing are crucial for efficient stem cell proliferation
X-Pure® gelatin outperformed standard research grade gelatins and other coating materials in 2D cell culturing
Irving, United States, 9th December 2021 / Sciad Newswire / Rousselot®, Darling Ingredients’ health brand and the global leader1 of collagen-based solutions, revealed the results of a new study which demonstrates that Rousselot’s ultra-pure GMP-grade2 gelatins outperformed standard non-endotoxin purified research grade gelatins and other coating materials in 2D cell culturing. Data demonstrate that endotoxin (lipopolysaccharide, LPS) levels and the nature of biomaterials used in cell coating have a significant impact on cell morphology and proliferation.
2D cell culture involves securing, nourishing, and growing cells on a flat surface. Most cells in the body are attached or anchored to an extracellular matrix (ECM) that consists of proteins like collagen, fibronectin, and laminin. Therefore, most cells require a coating with an ECM material that resembles their natural environment to improve cell attachment, proliferation, and sometimes cellular performance. A nourishing cellular environment means reducing factors that can lead to cellular stress. Multiple studies have shown that even traces of endotoxins can induce serious adverse effects on stem cell behavior and function3.
To secure and grow cells, Rousselot used gelatin, a biomaterial derived from collagen, to recreate the natural cellular environment within a laboratory setting. Rousselot investigated the effect of three different coatings on the morphology and proliferation of Adipose Derived Mesenchymal Stem Cells (AD MSCs):
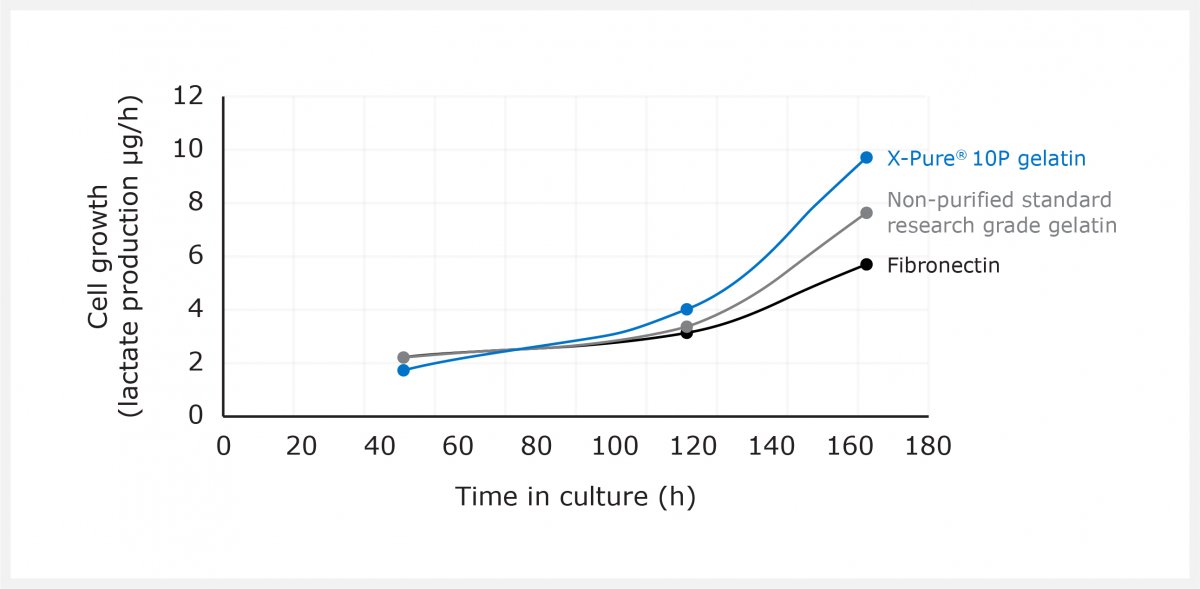
- Rousselot X-Pure® gelatin, characterized by low endotoxin levels (<10 EU/g) and 155 kDa molecular weight
- A commercially available standard research grade gelatin, with endotoxin levels of 4100 EU/g and molecular weight of 160kDa
- Standard fibronectin coating material, with endotoxin levels of 20000 EU/g and molecular weight of 220 kDa
“Stemness” of the cells may be better maintained on gelatin
The cell morphology of cells grown on discs coated with X-Pure or commercial gelatin showed elongated and aligned cells with a spindle shaped morphology, whereas the cells on fibronectin were less dense with more diversity in shape.
X-Pure low endotoxin gelatin best supported cell growth in 2D culture
In addition, Rousselot’s X-Pure gelatin coating resulted in approximately 70% increased cell growth (measured as lactate production during a 7-day culture period) compared to fibronectin. The research grade, non-purified gelatin coating only resulted in an approximately 35% increase (see graph).
“The use of low endotoxin gelatin should be standard practice in cell culture applications,” commented Jos Olijve, Scientific Support Manager at Rousselot. “Not only it is essential to nurture and promote cell growth to achieve consistent and reliable data, but as you move from basic cell therapy research to the clinic, low endotoxin products with complete documentation and traceability are essential for regulatory approval. Using purified, consistently produced, and high-quality biomaterials throughout product development minimizes the risk of unexpected and inconsistent results and speeds up translation.”
For more information on X-Pure or the impact of endotoxins on cell culture applications, visit the X-Pure Portfolio page.
Click here to view the full poster “Stem cell proliferation on 3D printed discs improved with low endotoxin and low molecular weight gelatin coating,” by Jos Olijve and Elien Gevaert Rousselot BV, Gent, Belgium.
References:
[1] Global Gelatin Market Insights Forecast to 2026, Calibre Research, 2020
[2] IPEC – Excipient Good Manufacturing Practices Guide, 2017 as of end 2021
[3] Magnusdottir A, Vidarsson H, Björnsson JM, Örvar BL. Barley grains for the production of endotoxin-free growth factors. Trends Biotechnol. 2013 Oct;31(10):572-80. doi: 10.1016/j.tibtech.2013.06.002. Epub 2013 Jul 10. PMID: 23849675.
ENDS
Notes to editors:
Rousselot® Biomedical
As the most recent strategic segment within Rousselot, we have drawn upon Rousselot’s 130+ years of worldwide expertise and proven track record of pharmaceutical gelatins and collagens to develop innovative ranges of purified, modified and non-modified gelatins and collagens for biomedical applications. Offering unique advantages to assure performance, quality, and safety from bench to clinic, Rousselot® X-Pure® and Rousselot® Quali-Pure™ provide consistent quality and are backed by strong scientific data and on-going research. Rousselot Biomedical is committed to support end-to-end partnerships to help “Advancing medical science”. https://www.rousselot.com/biomedical
Rousselot
Caroline Brochard-Garnier
Director of Communications & PR, Rousselot
[e] caroline.brochard-garnier@rousselot.com
Sciad Communications
Maria Patey
Communications Director, Sciad
[e] rousselot@sciad.com
Got an innovation you need bringing to life?
We’d love to hear from you! Head to our contact page to start a conversation.
